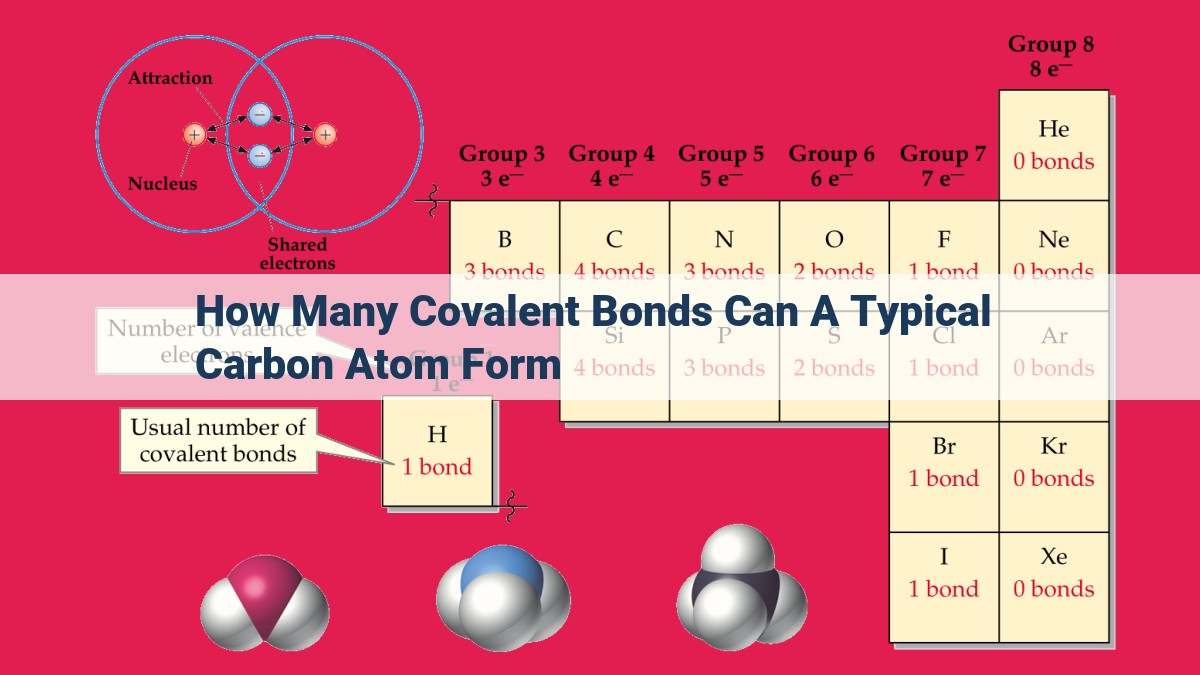
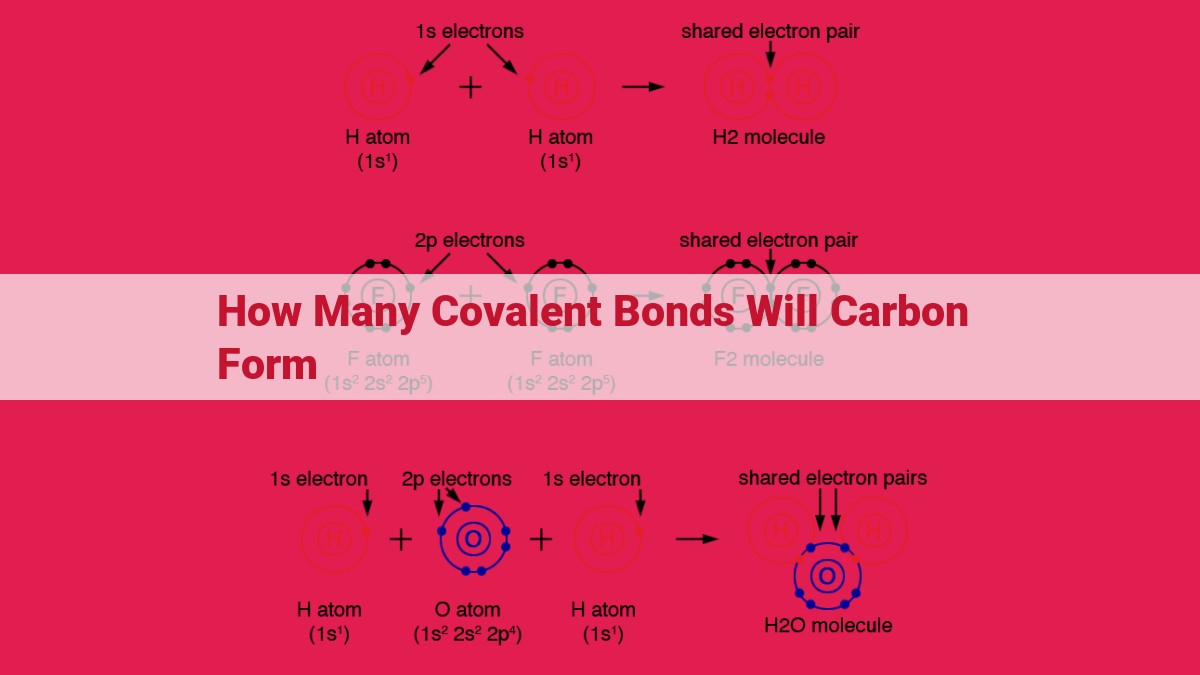
How Many Covalent Bonds Can Carbon Form - Since carbon has 4 valence electrons, it can form. It has 6 protons, 6 neutrons, and 6 electrons. A carbon atom can form four covalent bonds due to having four valence electrons. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. An atom can form covalent bonds by sharing its valence electrons with other atoms. This allows carbon to create various compounds, such. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. Carbon is a chemical element with the symbol c and atomic number 6. Of these 6 electrons, 4.
It has 6 protons, 6 neutrons, and 6 electrons. Of these 6 electrons, 4. An atom can form covalent bonds by sharing its valence electrons with other atoms. This allows carbon to create various compounds, such. Carbon is a chemical element with the symbol c and atomic number 6. Since carbon has 4 valence electrons, it can form. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. A carbon atom can form four covalent bonds due to having four valence electrons. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell.
An atom can form covalent bonds by sharing its valence electrons with other atoms. A carbon atom can form four covalent bonds due to having four valence electrons. Of these 6 electrons, 4. Carbon is a chemical element with the symbol c and atomic number 6. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. Since carbon has 4 valence electrons, it can form. This allows carbon to create various compounds, such. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. It has 6 protons, 6 neutrons, and 6 electrons.
PPT Organic Chemistry Functional Groups PowerPoint Presentation
Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. A carbon atom can form four covalent bonds due to having four valence electrons. Carbon is a chemical element with the symbol c and atomic number 6. Of these 6 electrons, 4. This allows carbon to share electrons.
MACROMOLECULES a.k.a. BioMolecules a.k.a. Organic Molecules ppt download
It has 6 protons, 6 neutrons, and 6 electrons. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. This allows carbon to create various compounds, such. An atom can form covalent bonds by sharing its valence electrons with other atoms. This allows carbon to share electrons with.
Carbon Compounds Section ppt download
A carbon atom can form four covalent bonds due to having four valence electrons. Of these 6 electrons, 4. It has 6 protons, 6 neutrons, and 6 electrons. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. This allows carbon to create various compounds, such.
Unit 2 (Biochemistry) Notes, Part 1 Atomic And Molecular Structure
An atom can form covalent bonds by sharing its valence electrons with other atoms. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. Carbon is a chemical element with the symbol c and atomic number 6. A carbon atom can form four covalent bonds due to having.
[Class 10 Chemistry] Bonding in Carbon Atoms Covalent Bonds
Carbon is a chemical element with the symbol c and atomic number 6. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. It has 6 protons, 6 neutrons, and 6 electrons. An atom can form covalent bonds by sharing its valence electrons with other atoms. Thus, a carbon.
The Chemistry of Carbon = Chemistry of Life ppt download
This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. Carbon is a chemical element with the symbol c and atomic number 6. Since carbon has 4 valence electrons, it can form. Of these 6 electrons, 4. It has 6 protons, 6 neutrons, and 6 electrons.
Mastering Carbon Bonds Understanding Hybridization and Covalent Bonding
A carbon atom can form four covalent bonds due to having four valence electrons. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. Since carbon has 4 valence electrons, it can form. This allows carbon to create various compounds, such. Of these 6 electrons, 4.
Part 2 Organic Chemistry (Carbon and Macromolecules) ppt download
Carbon is a chemical element with the symbol c and atomic number 6. Of these 6 electrons, 4. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. An atom can form covalent bonds by sharing its valence electrons with other atoms. A carbon atom can form four covalent.
Understanding Carbon's Bonding Properties for Stable Molecular Formations
An atom can form covalent bonds by sharing its valence electrons with other atoms. Carbon is a chemical element with the symbol c and atomic number 6. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. Since carbon has 4 valence electrons, it can form. This allows carbon.
PPT Introduction to Carbon Chemistry PowerPoint Presentation, free
A carbon atom can form four covalent bonds due to having four valence electrons. It has 6 protons, 6 neutrons, and 6 electrons. This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. An atom can form covalent bonds by sharing its valence electrons with other atoms. Carbon is.
Of These 6 Electrons, 4.
This allows carbon to create various compounds, such. Thus, a carbon atom can form a total of four covalent bonds with other atoms, as demonstrated in the methane molecule (ch4),. Since carbon has 4 valence electrons, it can form. A carbon atom can form four covalent bonds due to having four valence electrons.
It Has 6 Protons, 6 Neutrons, And 6 Electrons.
This allows carbon to share electrons with other atoms and reach the stable configuration of 8 electrons in its valence shell. Carbon is a chemical element with the symbol c and atomic number 6. An atom can form covalent bonds by sharing its valence electrons with other atoms.
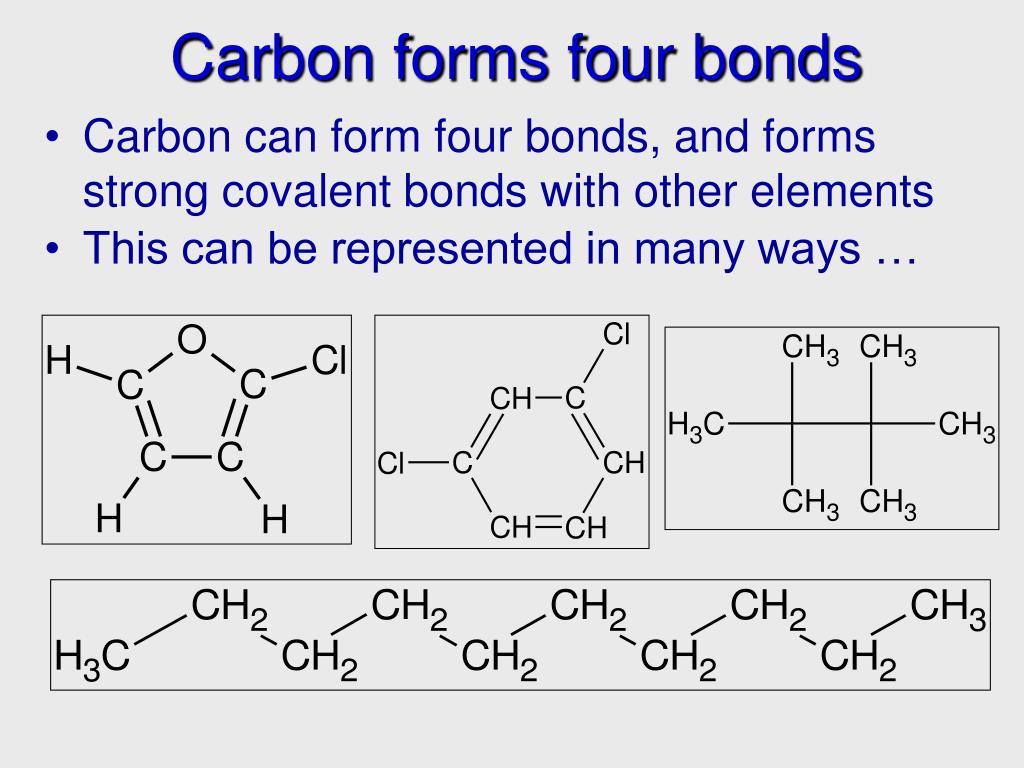



![[Class 10 Chemistry] Bonding in Carbon Atoms Covalent Bonds](https://d1avenlh0i1xmr.cloudfront.net/large/693c5ecd-56f0-42a1-aba9-2e5a5be31300/covalent-bonding-in-co2---teachoo.jpg)